 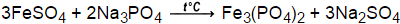 
One derivative is the glassy (i.e., amorphous) Graham's salt. The specific polyphosphate generated depends on the details of the heating and annealing. Polymeric sodium phosphates are formed upon heating mixtures of NaH 2PO 4 and Na 2HPO 4, which induces a condensation reaction. The cyclic polyphosphates, called metaphosphates, include the trimer sodium trimetaphosphate and the tetramer, Na 3P 3O 9 and Na 4P 4O 12, respectively. Of these salts, those of the diphosphates are particularly common commercially.īeyond the diphosphates, sodium salts are known triphosphates, e.g. In addition to these phosphates, sodium forms a number of useful salts with pyrophosphates (also called diphosphates), triphosphates and high polymers. Trisodium phosphate (anhydrous, hexagonal) Some of the most well known salts are shown in the following table. Three families of sodium monophosphates are common, those derived from orthophosphate (PO 4 3−), hydrogen phosphate (HPO 4 2−), and dihydrogenphosphate (H 2PO 4 −). Since safe and effective replacements for phosphate purgatives are available, several medical authorities have recommended general disuse of oral phosphates. Oral phosphate prep drugs have been withdrawn in the United States, although evidence of causality is equivocal. There are several oral phosphate formulations which are prepared extemporaneously. However, oral sodium phosphates when taken at high doses for bowel preparation for colonoscopy may in some individuals carry a risk of kidney injury under the form of phosphate nephropathy. Sodium phosphates are popular in commerce in part because they are inexpensive and because they are nontoxic at normal levels of consumption. They are also used in detergents for softening water and as an efficient anti- rust solution. They are also used in medicine for constipation and to prepare the bowel for medical procedures. They are also used to control pH of processed foods. For example, sodium phosphates are often used as emulsifiers (as in processed cheese), thickening agents, and leavening agents for baked goods. Sodium phosphates have many applications in food and for water treatment. The hydrates are more common than the anhydrous forms. Most of these salts are known in both anhydrous (water-free) and hydrated forms. Phosphate also forms families or condensed anions including di-, tri-, tetra-, and polyphosphates. 
Sodium dihydrogen phosphate Sodium hydrogen phosphate Trisodium phosphateĪ sodium phosphate is a generic variety of salts of sodium (Na +) and phosphate (PO 4 3−). H ope this helped, and that you were able to understand the procedure.For the chemical known as sodium phosphate, see Trisodium phosphate. All there is left to do is to divide these two numbers, and we would get our answer.Ġ.00376 mol Na 3 PO 4 = 0.15473 M Na 3 PO 4 (ANSWER!) Round it up to 0.155 to have 3 sig figs We just found out the number of moles of sodium phosphate, and we were given the number of liters of it. We know that Molarity = moles / Liters, hence I like to start doing conversion factors with the Liters we just got, from the side that has the most information given to us, which is Barium chloride, thusĠ.0564 L BaCl 2 x 0.100 Mol BaCl 2 X 2 mol Na 3 PO 4 = 0.00376 mol Na 3 PO 4 (These are the moles ofġ L BaCl 2 3 mol BaCl 2 Sodium phosphate that We also must know that 0.100M BaCl 2 can also be expressed as 0.100 mol BaCl 2 / 1 Liter. Hence, 56.4mL/1000mL will give us Liters, and 24.3mL/1000mL as well. I have always found it helpful to convert milliliters into Liters, when it comes to problems involving molarity. Information to find out: Molarity Sodium phosphate Not only does it makes it easier for us to answer, but when it comes to molarity, it is of vital importance to correctly balance it, otherwise we would never get the right answer! Here we go.ģ BaCl 2 (aq) + 2 Na 3 PO 4 (aq) -> Ba 3 (PO 4 ) 2 (s) + 6 NaCl(aq) Hello, and thank you for your great question! Thank you also for providingabalanced equation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
